Imagine sending viruses into orbit not to spread disease, but to learn how to cure it. That is essentially what a team of scientists did on the International Space Station, and the results may change how we think about infections on Earth.
Microgravity experiment on the International Space Station reveals surprising phage behavior
A new study led by researchers at the University of Wisconsin Madison asked a simple question. How do bacteria and the viruses that infect them behave when gravity almost disappears? In microgravity, fluids do not mix the way they do on Earth, nutrients drift slowly, and microbes live in a kind of weightless soup.
That strange setting turned out to be a powerful test bed for evolution.
In the experiment, the team packed test tubes with Escherichia coli bacteria and a virus called T7 bacteriophage, then flew them to orbit in a small hardware box that fit on a shelf the size of a laptop.
Astronauts incubated the samples for up to twenty three days, while a second set of identical tubes stayed on Earth as a control. The station is not sterile either. Microbiology work on board already tracks how everyday microbes hitch rides with crews and cargo.
How T7 bacteriophage and E. coli coevolved in space
At first, the viruses struggled in space. On Earth, T7 usually infects and destroys bacteria in less than half an hour. In orbit, infection was delayed for several hours, and both bacteria and viruses lost some viability during freezing and thawing.
Yet by the longest time point the phages had caught up and successfully wiped out most of their bacterial hosts. Genetic sequencing later showed that both sides had been mutating rapidly while they adapted to the new environment, something also seen in dedicated ISS microbiology research.
Those genetic changes did not look random. On the viral side, many mutations appeared in structural proteins that help the phage attach to the bacterial surface.
On the bacterial side, mutations clustered in genes that shape the outer membrane and help cells cope with stress, a theme that echoes other work on engineered living materials built with bacteria and fungi. In other words, even hundreds of kilometers above our heads, bacteria and phages kept playing their familiar evolutionary tug of war.
Space-driven mutations could help fight antibiotic-resistant infections on Earth
Back in the lab, the Raman lab took one more unexpected step. The team used the mutation patterns they saw in space to guide the design of new T7 variants on Earth, combining several of the beneficial changes into custom-built viruses.
When they tested these engineered phages against uropathogenic E. coli, the bacteria behind many urinary tract infections that often resist common antibiotics, some of the space-informed variants were far better at forming plaques and killing the pathogens than the original virus.
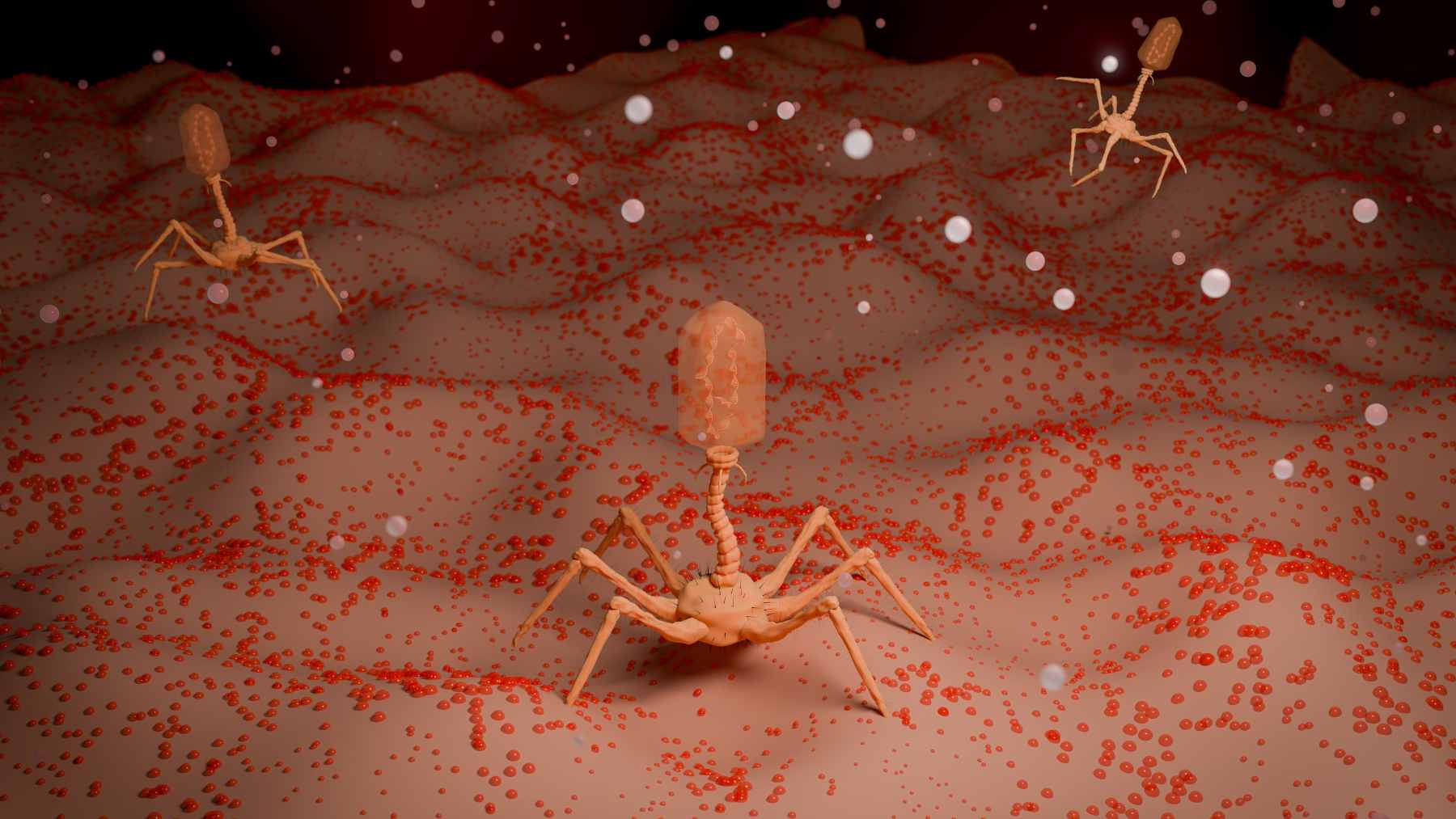
Why would a virus that learned its tricks in orbit help with problems in ordinary hospitals? The scientists suspect that the stressful conditions on the station, where nutrients and waste products move slowly and bacteria face unusual physical forces, may mirror the kinds of pressures microbes experience inside the human body, including dense biofilms in the gut.
That connects this work to other research on gut bacteria and how small shifts in microbial communities can have outsized effects on health.
Phage therapy and the growing crisis of antimicrobial resistance
The timing is no accident. Doctors are increasingly worried about antimicrobial resistant infections that shrug off standard drugs, including common urinary tract infections in clinics around the world.
Scientists are searching for new ways to fight superbugs, from space-trained phages to natural compounds such as the ant-derived chemicals that show promise against superbugs in the lab. Phage therapy has an extra advantage. Viruses can be tuned to target specific bacteria while leaving much of the microbiome intact.
What space microbiology teaches us about microbial ecosystems and sustainability
The experiment also hints at a broader ecological lesson. On Earth, bacteria adapt to pollutants, radiation, and climate stress in oceans, soils, and even radioactive sites, much like the newly described bacteria found in extreme environments.
In space, gravity itself becomes the stressor. Learning how microbial communities respond in this setting can help us predict how they will behave in closed habitats, from spacecraft to climate controlled vertical farms, and even guide future bio-based technologies such as living battery tech from microbes.
Future flights are expected to move beyond single virus and single bacterium setups. Researchers plan more complex experiments that mix several phages and bacterial species together, closer to a true gut microbiome.
Combined with ongoing microbial research on the station, this work could reveal new alliances and rivalries that shape life in closed ecosystems, whether those ecosystems orbit above us or sit in hospital wards and wastewater plants.
In the end, the story is surprisingly down to Earth. By watching tiny viruses and bacteria adapt in weightlessness, scientists are learning how to better defend human health in hospitals and homes, and maybe how to harness microbes as greener tools in a warming world.
The study was published in PLOS Biology.