Think about how often your bones quietly do their job. A quick walk to the store or carrying groceries up the stairs sends mechanical signals through your skeleton.
New research from Leipzig University in Germany suggests one little-known receptor may help turn those signals into stronger bone. In mouse studies, activating a receptor called GPR133 with an experimental compound increased bone strength and eased osteoporosis-like bone loss, pointing to a possible new path for future treatments.
A new target called GPR133
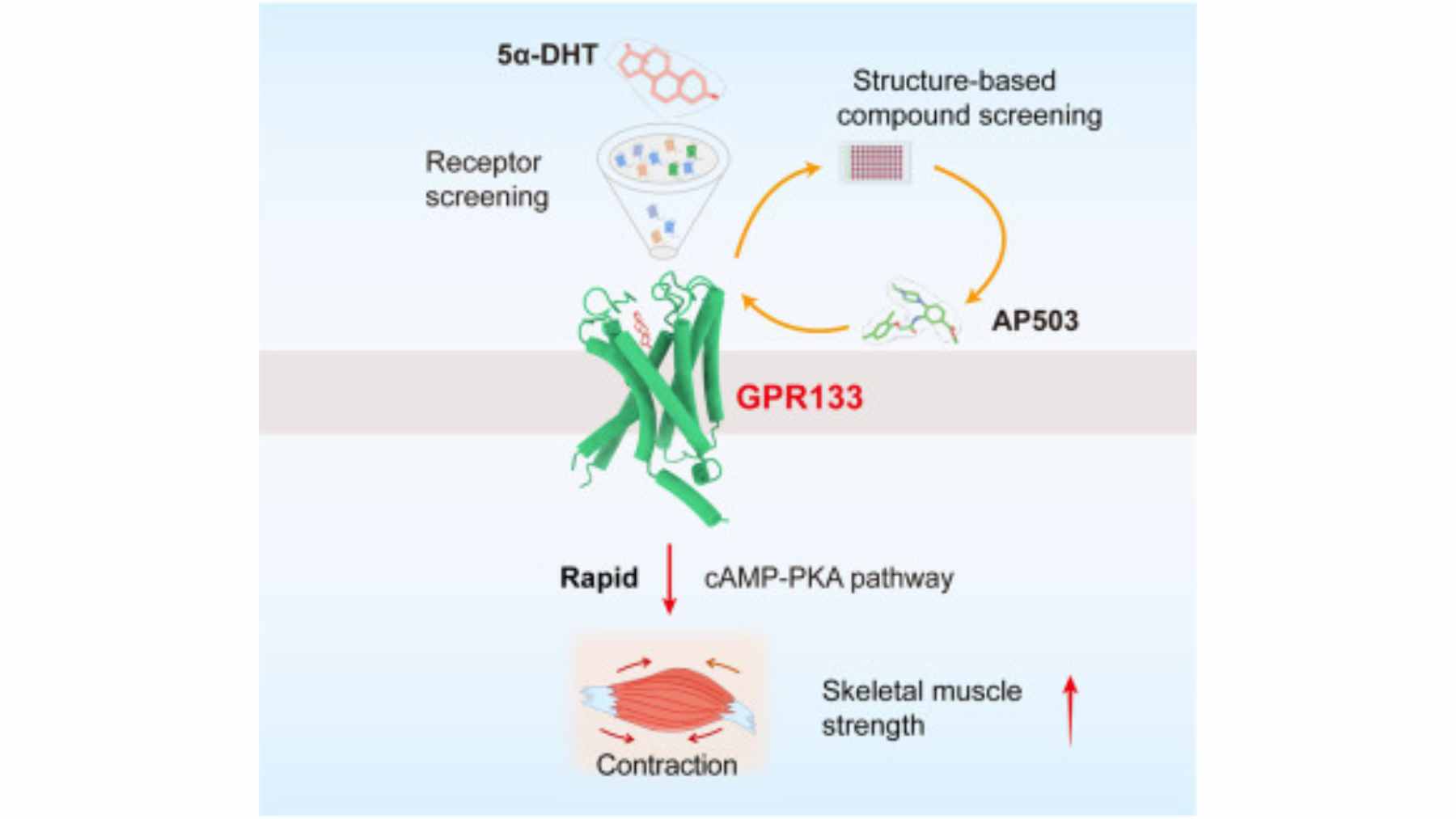
Researchers focused on GPR133, also known as ADGRD1, a member of the GPCR family that many medicines already target. Human genetic studies had linked variants in this gene to differences in bone mineral density and height, but the biology behind that link was still unclear.
To see what the receptor does, the team studied mice that lacked GPR133. Those animals developed lower bone mass and weaker bones in areas like the femur and the spine, a pattern the authors describe as characteristic of osteoporosis.
Then came the result that grabbed attention. An experimental compound called AP503, which turns on GPR133, “significantly increase[d] bone strength in both healthy and osteoporotic mice,” said Professor Ines Liebscher, the study’s lead investigator.
A disease that often stays hidden
Osteoporosis is sometimes called a silent disease for a reason. Many people do not realize their bones have become fragile until a low-impact fall or an awkward twist leads to a fracture.
In Germany, osteoporosis affects about 5.6 million people, and roughly 4.5 million of them are women, according to the International Osteoporosis Foundation. The same analysis estimates more than 831,000 fragility fractures occur each year in the country, which works out to about 95 broken bones per hour.
Zoom out, and the scale gets even bigger. The International Osteoporosis Foundation estimates that, worldwide, up to 37 million fragility fractures occur annually in people older than 55, which is about 70 fractures every minute.
Why “mechanical strain” matters
Bone is not a static scaffold. It is living tissue that remodels itself, and it pays attention to physical loading, the push and pull of daily life. Ever wonder why doctors keep talking about weight-bearing activity?
GPR133 appears to be one of the molecular “listeners” in that process. The paper reports that the receptor’s activity in bone-building cells depends on mechanical forces and contact with a partner protein called PTK7, which helps switch on signaling inside the cell.
In practical terms, that means a drug that nudges this pathway could be useful for people who cannot rely on movement alone, including those who are frail, immobilized after injury, or exposed to microgravity. The authors also note that exactly how mechanical and chemical activation work together still needs more study.
What AP503 did in an osteoporosis model
The researchers did not just test healthy animals. They also used an ovariectomy mouse model, a standard way to mimic postmenopausal osteoporosis, where estrogen loss accelerates bone breakdown.
In that model, activating GPR133 with AP503 improved multiple measures tied to bone health, including bone volume and mineral density, and it reduced the spacing that appears when trabecular bone thins out. It is a reminder that strong results in mice are not the same as a ready-to-prescribe therapy, but proof-of-principle matters.
The Leipzig team is also paying attention to muscle. In earlier work, they reported that stimulating GPR133 with AP503 strengthened skeletal muscle, raising the possibility of one treatment helping two age-related problems at once.
Where today’s treatments fall short
Modern osteoporosis care already has effective options, most of which work by slowing bone resorption. Common antiresorptive medicines include bisphosphonates and denosumab, while anabolic drugs such as teriparatide, abaloparatide, and romosozumab are used for some high-risk patients to help build bone.
The trade-offs are real, though. Bisphosphonate labels warn about rare but serious problems such as atypical femur fractures and osteonecrosis of the jaw, and some medicines have recommended limits on how long they should be used.
Some treatments also require careful planning around stopping or switching. Studies have found that discontinuing denosumab can be linked to a rebound rise in bone turnover and a higher risk of vertebral fractures, and the FDA label for romosozumab includes a boxed warning about potential cardiovascular risks.
What has to happen before people benefit
For now, this is early-stage science. The strongest evidence is in mouse models, and the researchers themselves point out that mouse and human bone biology differ in important ways.
Even so, discoveries like this get attention because the target looks drug-friendly. The study notes that GPCRs are among the most common molecular targets for medicines approved by the U.S. Food and Drug Administration, which could help if AP503-like compounds move toward human testing.
Leipzig University says its team is already running follow-up projects to explore AP503 in other diseases and to better map what GPR133 does across the body. That slow, methodical work is what turns a promising receptor into a therapy that is both effective and safe.
The environmental link hiding in plain sight
At the everyday level, the discovery is also a reminder that bones respond to movement. U.S. health agencies and bone-health organizations emphasize weight-bearing and muscle-strengthening activity as key tools for maintaining bone health, along with balance work to help prevent falls.
This is where ecology and health overlap in a surprisingly practical way. WHO has noted that walking and cycling can cut air pollution and help mitigate climate change, and it is hard to miss the difference between a brisk walk and sitting in traffic breathing exhaust fumes.
No lab-made compound can replace safe, appropriate movement for most people, and anyone worried about osteoporosis should talk with a clinician about screening and treatment.
The study was published in Signal Transduction and Targeted Therapy.