What if one of the most common metals on Earth could do a job we usually reserve for platinum, palladium, or other hard-to-source materials? Chemists at King’s College London say they have uncovered an unusual form of aluminum that hints at exactly that, at least in the controlled world of the lab.
The headline is simple but the implications are wide. A tiny triangle made of three aluminum atoms stays intact in solution and shows “unprecedented reactivity,” raising hopes for cheaper and more sustainable catalysts in the long run. It is exploratory research, but it lands right in the middle of today’s scramble for cleaner manufacturing and more resilient supply chains.
An aluminum triangle with outsized chemistry
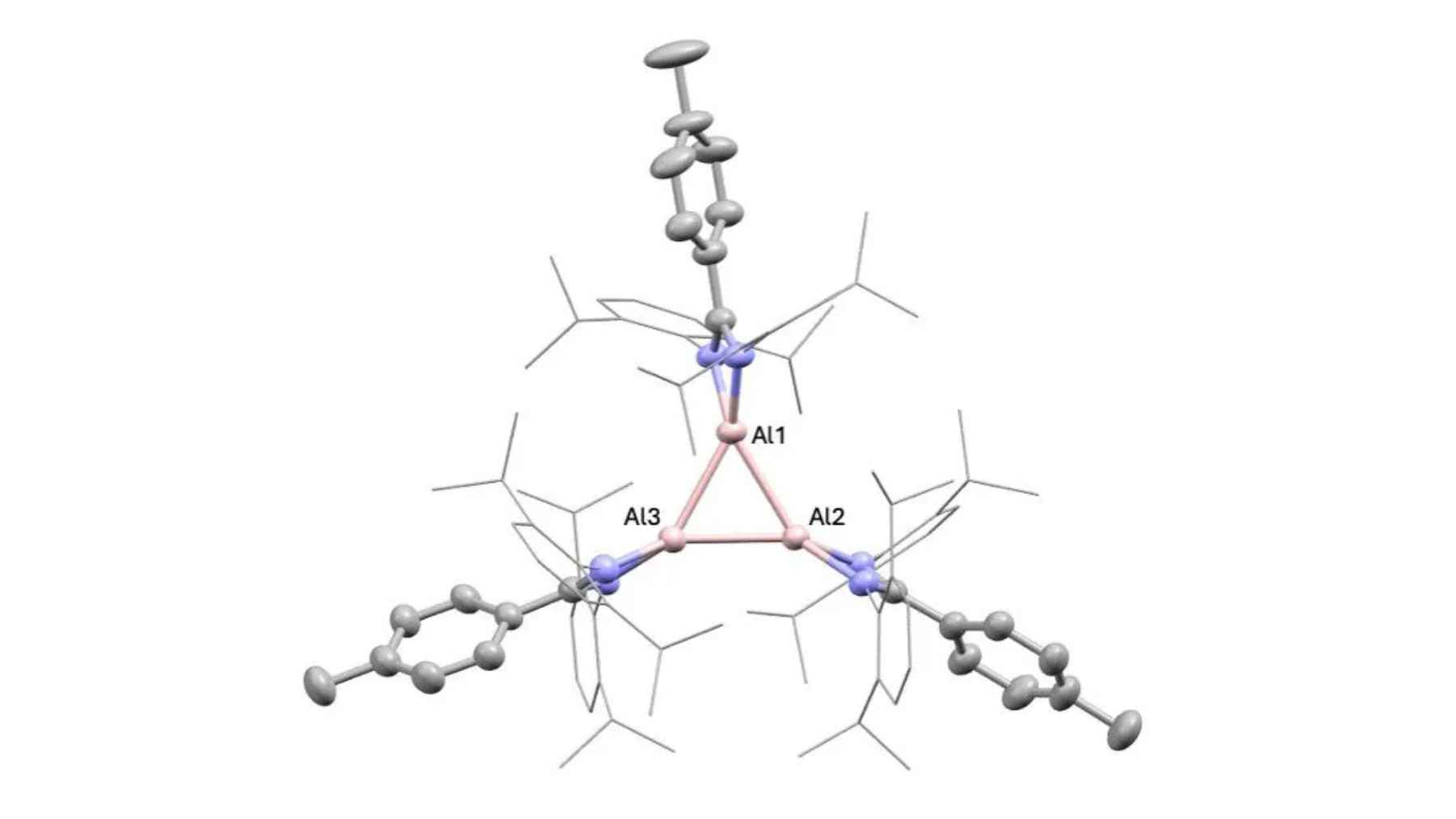
The new compound is a neutral aluminum(I) trimer called a cyclotrialumane. Think of it as a three-atom ring of aluminum that behaves less like the familiar metal in foil and cans and more like a highly reactive chemical tool. (nature.com)
That structure matters because, as the Nature Communications paper explains, neutral trimeric aluminum(I) structures had been “notably absent” from earlier work. The researchers report two examples, and they show the trimeric form is retained in solution, which makes it available for a broader set of reactions.
King’s College London describes the compounds as able to “break apart tough chemical bonds” while also revealing molecular structures “never been observed before.” The team argues this is not just imitating transition-metal chemistry, but pushing into new territory that those metals may not reach easily.
Why catalysts are a climate story
Catalysts sit behind the scenes of modern life. They help make bulk chemicals, fuels, plastics, and medicines faster and with less waste, and they often determine how much heat and electricity a process needs. When a factory reaction runs cooler and cleaner, that ripple can show up everywhere, from lower emissions to lower costs.
But many of the best-known catalysts rely on expensive metals that are environmentally damaging to extract and can be hard to secure. Bakewell’s group points to the supply problem directly, noting that “transition metals are the workhorses of chemical synthesis and catalysis” but many are “increasingly difficult to access and extract.”
Aluminum is appealing because it is abundant. In the King’s release, Bakewell says, “We chose aluminium as it’s super abundant, making it about 20,000 times less expensive than precious metals such as platinum and palladium.” If researchers can translate that advantage into real catalysts, it could reduce pressure on some of the most disruptive mining and refining chains.
Critical minerals are getting harder to ignore
This discovery is not about rare-earth magnets, but it fits a broader trend. The U.S. Geological Survey estimates global rare-earth mine production in 2024 at about 430,000 short tons (390,000 metric tons) of rare-earth-oxide equivalent, with China producing about 298,000 short tons (270,000 metric tons).
In the same USGS summary, the United States produced about 49,600 short tons (45,000 metric tons) of REO in mineral concentrates in 2024, valued at $260 million. It also reports that catalysts were the leading domestic end use of rare earths, while many magnets arrive embedded inside finished products rather than as raw materials.
Markets and policy are reacting to this pressure. Reuters reported that neodymium-praseodymium oxide prices jumped in 2025, rising from about $29 per pound ($63 per kilogram) to about $40 per pound ($88 per kilogram) after supply disruptions. When key inputs become volatile, labs start looking even harder for substitutes.
What the experiments found
In Nature Communications, the authors report that the cyclotrialumanes can activate small molecules and unsaturated substrates, including hydrogen, benzene, and an alkyne. That kind of bond activation is the starting point for many industrial reactions, even though these early tests are not yet a commercial catalytic system.
Some reactions proceed quickly under relatively mild conditions. The paper describes rapid reaction with hydrogen at about 1 bar, which is roughly 15 pounds per square inch, and room-temperature behavior that is easy to follow by a sudden color change in solution. With ethylene at similar pressure, the trimer reacts immediately and forms new aluminum-carbon structures.
The ethylene chemistry is where the “triangle” really earns its keep. The authors describe 5- and 7-membered aluminum-carbon ring systems formed through a series of reactions, and they note these products are “without precedent” for both transition metals and main-group metals.
It is an example of how a small, stable cluster can unlock reaction pathways that a single metal atom might not access.
What comes next for greener chemistry
The biggest test is whether this reactivity can be turned into practical catalysis. A reagent that reacts once is impressive, but industry needs systems that can run many cycles, stay selective, and handle messy real-world mixtures without falling apart.
There is also an honest sustainability check to make. Aluminum is abundant, but upstream production can create significant waste, and the U.S. EPA notes that making aluminum generates about 2 to 2.5 tons of solid waste for every ton of aluminum produced, including red muds that can pose environmental risks. Cleaner catalysts still benefit from cleaner power and better waste handling at the source.
Bakewell emphasizes that the work is early, saying the team is “very much in the exploratory phase” and “just at the start” of unlocking what these materials can do. Still, the direction is clear, and it is hard not to wonder where else common elements could surprise us.
The original press release was published on King’s College London.