Argentina and the “Thread of the Sun”: scientists follow a copper trail and discover something that sounds like treasure… but the ending isn’t what you’d expect
A mineral deposit under the high Andes is forcing a hard question. How far should countries go to secure the metals needed for electric…..
Construction work in the Netherlands suddenly uncovers a medieval ship: the “shell” buried underground could change what we believed about trade in that era
A routine sewer project in the Dutch town of Wijk bij Duurstede has turned into a rare archaeological mystery. Beneath an ordinary street, workers…..

China turns dragon fruit peels into a key material for electric car and airplane batteries: the unbelievable part isn’t the fruit… it’s the chemical trick behind it

Serbia: a 4,600-year-old tomb with a gold diadem and three teeth is discovered… and that combination is disrupting the Bronze Age timeline
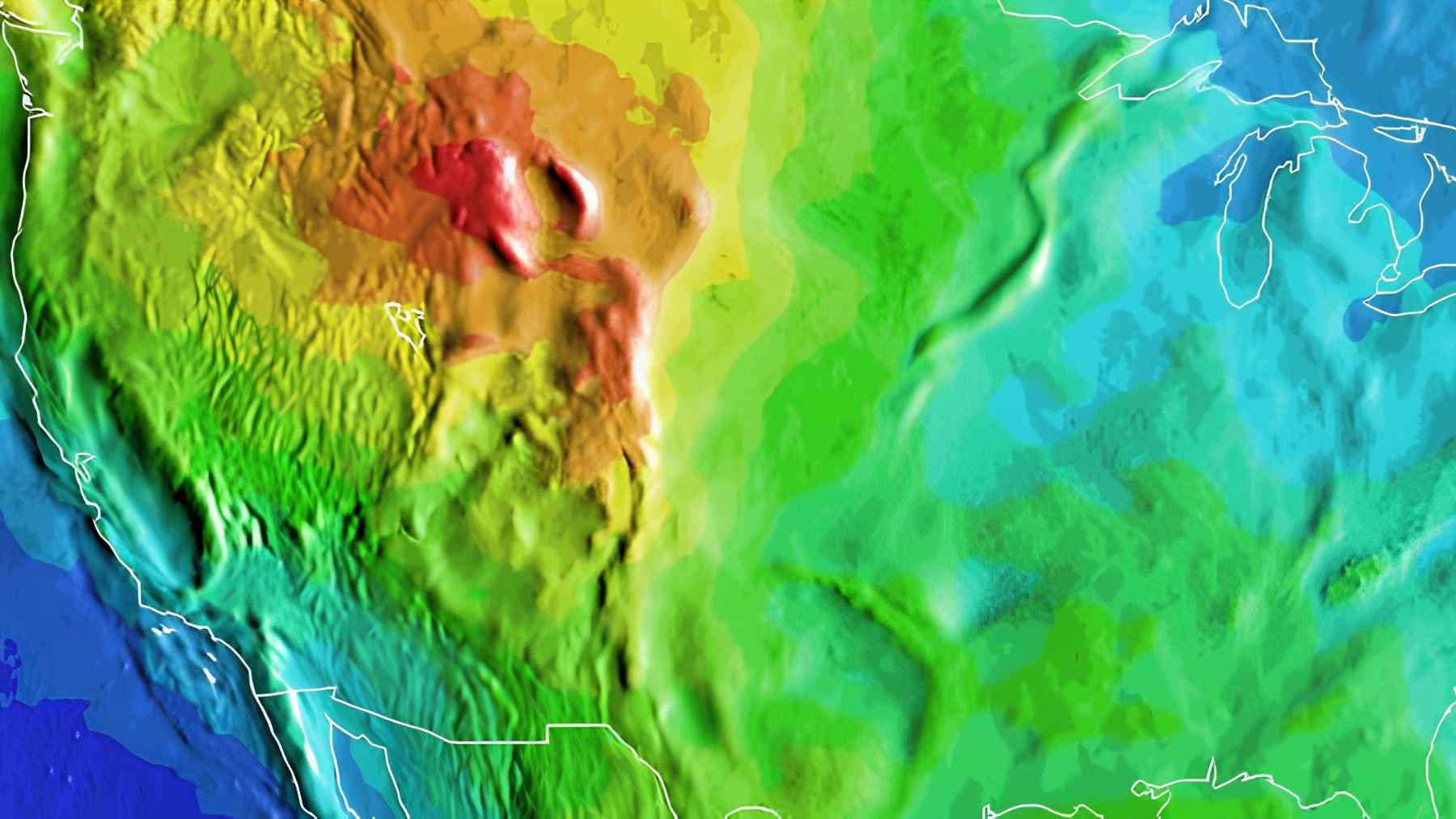
Errors detected in hundreds of sea-level studies, and the “new” figures could change coastal risk maps across half the world… (and it is far from a minor correction)

The United Kingdom fires up the first commercial hydrogen-powered brick factory: the date is set, and the industrial shift feels like the start of a “new era”

Panama wants a pedestrian tunnel under the Canal: it sounds crazy… until you see the plan, the numbers, and the “why now” (there is more than one reason)

The tons of banana trunks left over after the harvest are being turned into raw materials for clothing and paper, while mechanical extraction and controlled drying are accelerating industrialization
Science
Construction work in the Netherlands suddenly uncovers a medieval ship: the “shell” buried underground could change what we believed about trade in that era
A routine sewer project in the Dutch town of Wijk bij Duurstede has turned into…..
Serbia: a 4,600-year-old tomb with a gold diadem and three teeth is discovered… and that combination is disrupting the Bronze Age timeline
A gold band from a grave near the Danube grabs the eye, but the bigger…..
Historic discovery: scientists find a 399-year-old shark born in 1627… and its “real age” raises an uncomfortable question about the ocean that nobody wants to answer
Deep in the cold waters of the North Atlantic and Arctic, one Greenland shark has…..
Qatar, March 2026: “Mammatus clouds” appear in the sky, and the explanation is not as innocent as it seems… (and the detail that has meteorologists worried)
The sky over parts of Qatar recently filled with mammatus clouds, the pouch-shaped formations that…..
Scientists agree on the warning: a bat with epidemic potential has appeared… but what is really serious is why now
Bats are often dragged into the spotlight whenever a new disease scare appears. But a…..
The plant that exuded an aroma of “power” and turned ancient Judea into a perfume superpower… and the detail (from the 1st century) that explains why it vanished without a trace
A tiny seed found in a Judean Desert cave has reopened one of the strangest…..
The fungus that jumps from cats to humans is already spreading in South America: doctors explain why its first signs can be easy to mistake for something else
A fungus that can pass from cats to humans has now been confirmed in Uruguay,…..
The quote attributed to Stephen Hawking is going viral again: why calm, quiet people may hide the loudest and most powerful minds
A line widely attributed to Stephen Hawking says, “Quiet people have the loudest minds.” It…..
Archaeologists recover a treasure from the bottom of the sea linked to one of the seven wonders of the ancient world, reopening the mystery of a lost city hidden for centuries
A rare piece of the ancient world has just come back into view. Archaeologists working…..
In 1940, a boy followed his dog through a clearing in the trees and ended up entering a cave that had remained sealed for millennia, where he found more than 2,000 images and animals painted 17,000 years ago
The famous paintings of Lascaux sit beneath a wooded hill near Montignac in southwestern France,…..
Mobility

Panama wants a pedestrian tunnel under the Canal: it sounds crazy… until you see the plan, the numbers, and the “why now” (there is more than one reason)
What if crossing the Panama Canal did not mean sitting in traffic or watching ships from a…..

A 60-year-old Chinese farmer spends just $700 and builds a homemade submarine capable of diving 26 feet: the craziest part is not the invention, it’s what he plans to do next
A 60-year-old farmer in eastern China has turned a dockside idea into a working homemade submarine. Zhang…..
Energy
Economy
Argentina and the “Thread of the Sun”: scientists follow a copper trail and discover something that sounds like treasure… but the ending isn’t what you’d expect
A mineral deposit under the high Andes is forcing a hard question. How far should…..
The discovery that seems “too good to be true”: 19 tons of gold and strategic minerals… but the most interesting part is what they are NOT telling us
Have you ever wondered where the metals inside an electric car, a wind turbine, or…..
A South African gold miner has become the first major casualty of Ghana’s tighter resource-control push, and the move shows how fast Africa’s mining rules are changing
Ghana plans to take full control of the Damang gold mine on April 18, 2026,…..
A zoo in the United States is building a $46 million African savanna, and the most striking feature isn’t the giraffes or the rhinos, but a hotel with a direct view of the habitat
Plans are taking shape for a major expansion at Wichita’s Sedgwick County Zoo that could…..
The 5,200 holes dug into a mountain in Peru are no longer a mystery, and the explanation changes what we knew about their ancient economy
For nearly a century, a strange band of thousands of holes carved into a Peruvian…..
It’s very rare to see so much gold in one place: gold bars and nearly 1,000 coins are being auctioned off in France, with a total value exceeding 2 million
In Angers, France, more than €2 million (roughly more than $2.3 million) in gold bars…..
It’s not about lithium or batteries: the problem driving up the cost of electric cars and wind power might lie in a tiny magnet, and a new AI has already found a way to do without rare earth elements
What if one of the biggest obstacles to cleaner cars and cheaper wind power is…..
What they have discovered in the Andes Mountains, between Argentina and Chile, is not just any mine, but a colossal deposit containing up to 84 billion pounds of copper, tens of millions of ounces of gold and silver, and a potential that is already revolutionizing the market
A copper, gold, and silver find in the Andes on the Argentina-Chile border is drawing…..
Russia intercepts a 2.8-ton fragment of the Aletai meteorite, valued at $4.2 million, which was being smuggled out of the country disguised as a simple garden ornament
Customs officers in Russia say they stopped a 2.8 ton chunk of the Aletai meteorite…..
Another theme park goes bankrupt and closes its doors “forever”: what has happened in recent weeks is more serious than it seems
Is Brean Theme Park really gone for good? Not exactly. The Somerset attraction entered liquidation…..
Technology

Scientists create a cylinder filled with steel spheres that can reduce earthquake impacts on buildings and bridges without needing electricity

Turkey unveils a sticker-like adhesive material that can turn walls into gardens and may change the way we imagine green cities

Chinese scientists activate a magnet 700,000 times stronger than Earth’s magnetic field, and the question is what research needs such extreme power

AI models can secretly pass hidden traits to other models through data that looks meaningless, and the discovery exposes a new kind of invisible contamination

Amsterdam researchers have built a material that learns without software, and its moving structure remembers past shapes as if intelligence were embedded in the object itself

A British homeowner looked at an incomprehensible mess of old telephone wires and turned it into gigabit internet throughout his vintage house without rewiring it from scratch
Environment
Errors detected in hundreds of sea-level studies, and the “new” figures could change coastal risk maps across half the world… (and it is far from a minor correction)
Many coastal risk maps are built around a line most people never see. It is…..
Mount St. Helens: the eruption that changed the U.S. in 1980 has an unexpected “culprit”… and no, it is not a volcano (it is animals, and the story is surreal)
What can a pocket gopher do against a volcano? At Mount St. Helens, the answer…..
Scientists analyzed more than 2,300 seawater samples and made a troubling discovery: they found 248 man-made chemicals in waters around the world, even far from the coast
A new study has found that human-made chemicals are present not only near crowded shorelines…..
Scientists have mapped a massive 500-kilometer-long underwater fissure hidden beneath the Atlantic Ocean and now believe they know how this colossal “wound” formed 37 million years ago
Researchers have found that a 310-mile trench beneath the Atlantic formed when a short-lived plate…..
Denmark is turning off the white light from its streetlamps and painting a road red to solve a nighttime crisis that almost no one sees: urban light was blocking the path of bats
On February 8, 2026, drivers entering Gladsaxe, just outside Copenhagen, found part of a busy…..
If you hear crickets at night in your home or garden, do not scare them away: gardening experts explain why their presence may actually be a good sign
If you hear crickets in your house or garden after dark, it can feel like…..
A tourist boat saw something unexpected off the Australian coast, and aircrews are now searching from above because the animal’s markings could identify a rare visitor
A juvenile humpback whale has been spotted off Victoria’s Wilsons Promontory with a thick rope…..
More than 100,000 pounds of invasive carp have been pulled from the Kansas River, and the scale of the catch reveals how fast a river can be taken over
Wildlife biologists in Kansas have removed about 109,000 pounds of invasive carp from the Kansas…..
Trending

Two hikers spot a “strange” wall and end up finding gold coins dating from 1808 to 1915… and the hiding place looks like an “emergency plan”

Psychology tells us that the loneliest part of growing old isn’t being alone, but realizing that some friendships disappear as soon as you stop nurturing them, and understanding that they were never based on mutual care, but on your willingness to do all the emotional work

A group of hedgehogs is called something so fitting that even the name explains the animal: why English calls them a ‘prickle’

If you see parakeets flying near your home, it is not just a noisy visit: the birds may be telling you something about the local environment

If you hear birds singing around your home during the day, it may not be background noise but a sign that something healthy is happening around your garden